AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Regeneron stem cell2/28/2024 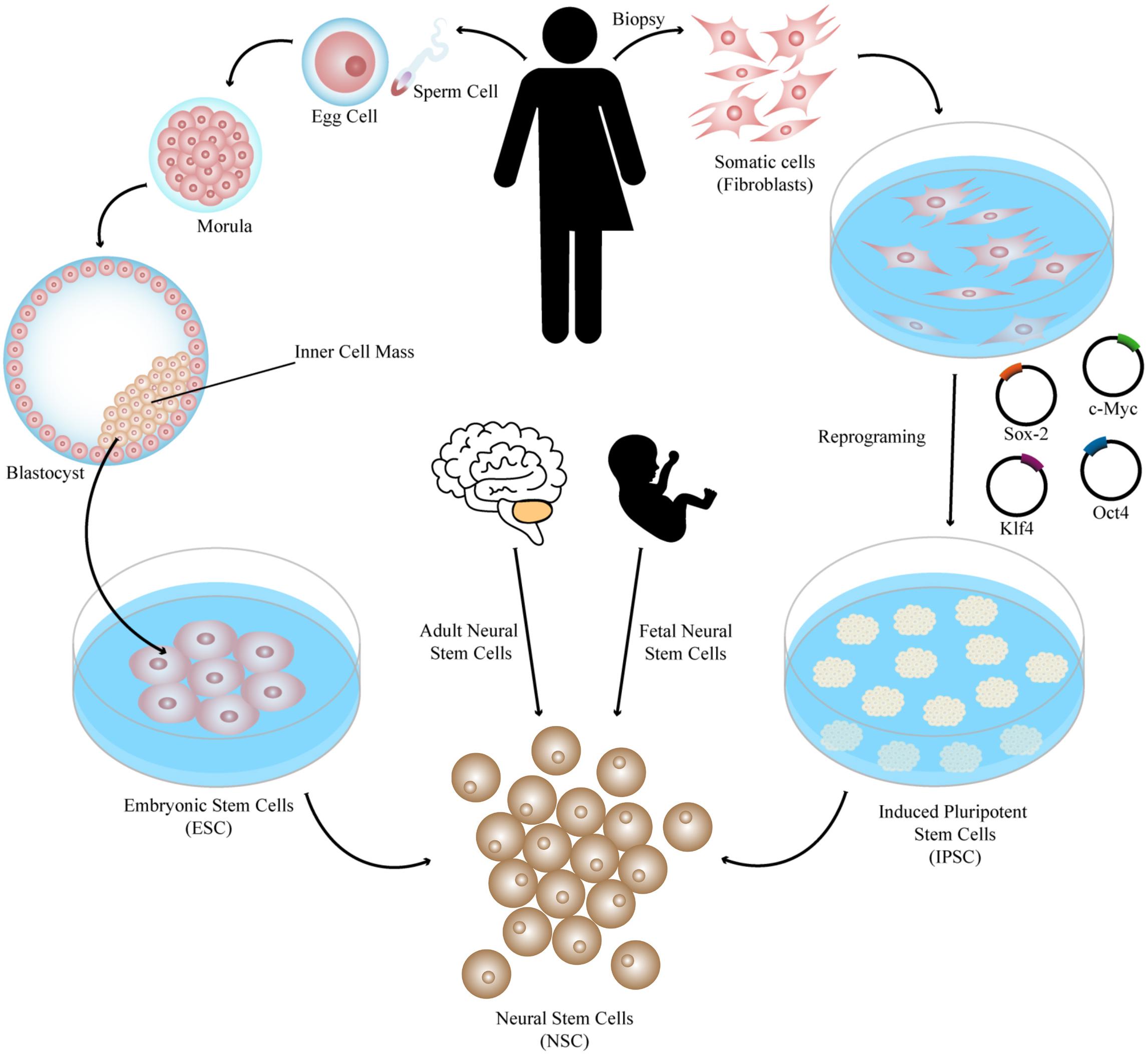
The positive opinion is supported by results from the global Phase 3 EMPOWER-Lung 3 trial, which was designed to closely resemble a patient population with varied disease presentations that physicians manage in everyday clinical practice. Food and Drug Administration (FDA) for advanced NSCLC regardless of PD-L1 expression. In November 2022, the Libtayo combination was approved by the U.S. The European Commission is expected to make a final decision on the application in the coming months. This includes patients that have no EGFR, ALK or ROS1 aberrations and whose tumors are metastatic or locally advanced and not candidates for definitive chemoradiation. Specifically, the CHMP recommended the Libtayo combination be approved in the European Union (EU) for first-line treatment of adult patients with advanced non-small cell lung cancer (NSCLC) with ≥1% PD-L1 expression. (NASDAQ: REGN) today announced the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion for Libtayo ® (cemiplimab) in combination with platinum-based chemotherapy. 24, 2023 (GLOBE NEWSWIRE) - Regeneron Pharmaceuticals, Inc. 
Recommendation based on a Phase 3 trial demonstrating superior survival outcomes for Libtayo plus chemotherapy compared to chemotherapy alone
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
